PLATELET GEL VIRTUES
A full description of the mechanisms leading to the platelet-induced tissue-healing and tissue-regeneration is almost impossible. However this figure (taken from a recent paper published by Everts et al. JECT 2006;38:174–187) depicts the general healing process induced by platelets.
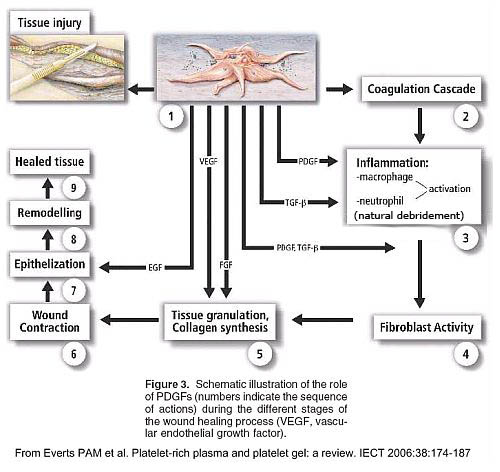
These mechanisms are at work whatever the tissue to be healed, repaired, or regenerated. This is the reason why platelets exert their regeneration activity on virtually all tissues (skin, bone, cartilage, tendon, muscle, myocardium, ligament, and so on).
In addition to the major healing effects deriving from platelet-derived factors, three additional relevant aspects are worth to be mentioned. The antibacterial effect; the reduction of the general costs of the patient management; the safety of the product.
Antibacterial effect
Haemostatic property apart, platelets have conserved through evolution some natural immunologic and bactericidal functions. In particular it was demonstrated that secreted platelet antimicrobial proteins are bactericidal to blood borne pathogens. However, taking this as a fact, one must not underestimate that some environmental bacteria can be insensitive to the platelet antimicrobial proteins. Furthermore, one must consider that the gel itself might function such as a bacterial growth medium. In conclusion, the platelet gel exerts bactericidal properties but it must be handle as far as possible in clean (sterile) conditions.
Reduction of the management costs
The application of platelet gel reduces the tissue-healing time. For the recovered inpatient subjects the length of hospital stay is shortened; in transfusion-dependent surgery transfusion requirements and post-operative morbidity are reduced; for the outpatient subjects the recovery time is shortened as well; the prevalence of the tissue-healing complications is reduced. This leads to every kind of direct and indirect care-related costs (hospital’s costs; national health care’s costs; nursing and dressing costs; invalidity or lower working capacity-related costs; complication-related costs) to be reduced. For example, a study on the sternal wound infection-related costs in a cardiosurgery unit demonstrated $ 500,000 saving per year using prophylactic platelet-gel dressing of the sternal wound versus $ 1,200,000 overheads without platelet-gel treatment. It has also been maintained that, in some countries, cost saving is obtained treating the diabetic foot with high-cost recombinant human growth factor (Ghatnekar et al. Pharmacoeconomics 2001; 19: 767-778). Moreover, using a low-cost growth-factor therapy such as that provided by Plateltex® the cost saving might be significant. Considering that, only in the US, there are over 100 million surgeries performed annually and roughly 7 million people suffer from chronic, considerable resource conservation might be realized extending the therapeutic and prophylactic use of platelet gel.
Safety
The safety of the autologous gel depends on the autologous source of the platelet rich plasma. This eliminates the concern of blood-transmissible diseases and of immunologic reactions. Rare reactions to bovine thrombin have been described. Bovine thrombin is no more marketed EU countries. Plateltex® provides batroxobin as gel forming activator. Despite the fact that the growth factors have strong mitogenic properties there is no evidence that the platelet gel promote tumor growth or carcinogenesis. It must be stressed that there is some evidence that batroxobin itself exerts antimetastatic effects (Shibuya M et al. J Cancer Res Clin Oncol 1990;116:168-172).
In any case the growth factor therapy is contraindicated in lesion with signs or at risk of neoplastic degeneration.
A fundamental prerequisite for the platelet gel efficacy, is the intrinsic quality of the platelet concentrate which is activated to produce the gel. Relevant factors are the platelet concentration and the growth-factor content. In the platelet concentrates, the platelets must be 4-8 folds the platelet concentration measured in the whole blood prior processing. This means that the measured platelet concentration must be 800,000/2,000,000 platelets per microliter. During the procedure to get such platelet count, platelets must not undergo excessive mechanical stress, since tissue-effective growth factors are lost during the procedure if platelets are treated under stressing conditions. Plateltex® provides a method which meets these requirements.